Out-gassing from water, resins, and core materials behaves like a vacuum leak and can severely limit achievable vacuum levels during infusion.
Vapour Pressure
Evaporation and sublimation into a vacuum are collectively referred to as out-gassing. All materials—solid or liquid—have a finite vapour pressure, and out-gassing becomes significant when the surrounding vacuum pressure drops below that vapour pressure. In man-made vacuum systems, out-gassing has the same practical effect as a leak and can limit the achievable vacuum level.
Water vapour is very often the unseen cause of vacuum pump problems. It originates from moisture present in the atmosphere, which is absorbed into fabrics and onto the surfaces of moulds and bagging films. Under certain combinations of vacuum level and ambient temperature, as shown in the water boiling chart, this moisture will evaporate and be transported to the vacuum pump.
Especially when the pump is cold, water vapour will condense into liquid water on the exhaust side of the vacuum pump. This liquid water may circulate with the pump oil and can damage the pump in several ways. It will re-evaporate on the inlet side, occupying a large volume and reducing the effective pumping capacity. When the pump is stopped for any length of time, liquid water inside the pump promotes internal corrosion.
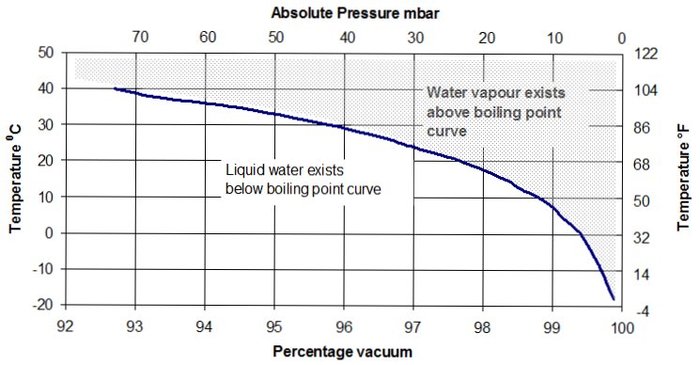
Some amount of water vapour is present in almost every vacuum situation, but a dramatic increase in vapour volume occurs once liquid water begins to boil. Water boils at progressively lower temperatures as the vacuum level increases. Any combination of temperature and pressure above the boiling-point curve will generate large volumes of water vapour until all available water has evaporated.
During this process, a noticeable reduction in vacuum pump performance should be expected until the pump has removed the water vapour and expelled it through the exhaust.
Another source of vapour is resin solvents. Polyester and vinylester resins in particular will out-gas under vacuum. The vacuum level at which this occurs depends on the vapour pressure of the solvents involved. Resin suppliers can provide guidance on the vapour pressure of their formulations and the maximum vacuum level that should be applied after infusion to avoid unnecessary solvent out-gassing that could interfere with the curing process.
This is one of the reasons why specially formulated vacuum infusion resins are used— not only for their low viscosity, but also to minimize out-gassing issues. Most epoxy resins are relatively safe in this respect.
Some foam core materials are also known to suffer from out-gassing problems, which is one of the reasons I chose Corecell as the core material for my composite parts and boat hulls.
To minimize problems caused by water vapour, raw materials should be kept dry during storage, preferably in a dehumidified environment, and the work area should be kept as dry as practical. To avoid resin vapour problems, use a suitable infusion resin and regulate the vacuum level after infusion by maintaining a vacuum slightly poorer than the vapour pressure of the resin solvent.
If the achievable vacuum level appears abnormally poor despite careful leak sealing, high vapour pressure is a likely cause. When checking for leaks, always perform pressure-rise or leak-back tests at a vacuum level poorer than (or an absolute pressure higher than) the boiling-point pressure.
For example, at an ambient temperature of 30 °C (86 °F), leak testing should be carried out at a vacuum poorer than about 95% or an absolute pressure higher than roughly 50 mbar (≈ 0.73 psi or 1.5″Hg — and yes, I still hate conversions; please consult convert-me.com, my favourite online unit converter).
Food for thought: simple numerical converters cannot account for the difference between gauge vacuum and absolute pressure. On convert-me.com/pressure, 0 mbar equals 0″Hg, whereas—as you now know— 0 mbar absolute corresponds to 29.92″Hg (at sea level in a standard atmosphere).